What is the BEYOND-9 clinical research study?
If you live with hemophilia B, please know that you are not alone in your medical journey. Hemophilia B is a rare genetic disease that affects about 4 in every 100,00 males worldwide according to the World Federation of Hemophilia (WFH).
Currently, we are working on developing potential treatments for hemophilia B. BEYOND-9 is a
phase 1/2A phase 1 clinical research study is a clinical research study investigating the overall safety and tolerability of an investigational treatment. In a phase 2 clinical research study, investigators assess the safety of the potential treatment and the best dose. Researchers also study how it works and possible side effects. clinical study that aims to see if an investigational CRISPR-based targeted gene insertion therapy may be effective and well-tolerated in males with hemophilia B. This treatment is called “investigational” as it has not yet been approved to treat hemophilia B.
Am I eligible?
What is the study treatment?
What can I expect if I participate?
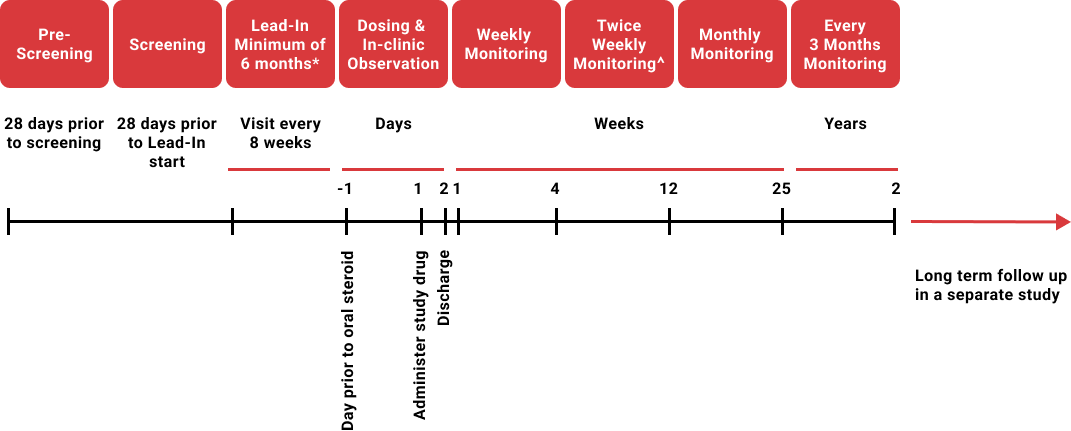
Study flow diagram